 The idea behind an atomic bomb is really very simple: bring lots of uranium-235 together very quickly so that it undergoes very rapid fission. An atomic bomb can be thought of a very rapid meltdown. More and more neutrons are released hitting more and more U-235 atoms, producing so much heat that everything around the uranium melts. A meltdown is when there is a runaway fission chain reaction. Not only had the scientists (lead by the brilliant Italian, later American, physicist Enrico Fermi) created the world's first self-sustaining nuclear reactor, but had the last brick dropped into the "pile" they would have been witness to the world's first nuclear meltdown.Īt the end of the previous section the word "meltdown" was used. However, at the time no one was really sure of the full health effects of radiation exposure and with World War II raging the safety of individuals was second to carrying out what was seen as vitally important work. Looking back on this event it is almost unbelievable that the experiment was allowed to go ahead. The insertion of the last brick was just enough to start a controlled chain reaction in the U-235 as more and more neutrons were released as a result of nuclear fission. Graphite bricks containing U-235 were piled up in a box shape and a further single brick was gradually inserted into the box. This was first done at the end of 1942 in a disused squash court under the University of Chicago's Stagg Field stadium. Although the half-life of U-235 is a very long time, if we get enough of the atoms together in one place the chances that any one of them will undergo spontaneous fission is very, very high. If we do that we don't even need to supply the first neutron. Firstly, we see a neutron striking a uranium-235 atom to make a uranium-236 atom:Īll we have to do is get enough U-235 in one place. After a short while the uranium-235 splits into an atom of barium-141 (Ba-141), an atom of krypton-92 (Kr-92) and three neutrons. First we fire a neutron (n) at the uranium-235 (U-235) atom so that it sticks to it. We will look at the most common one that of uranium-235 splitting into barium, krypton and three neutrons. there are many different particles that can result from the decay. 
There are many different "channels" through which uranium- 235 can decay, i.e. This is a very unstable isotope and decays rapidly by splitting into lighter atoms and particles. However, it was discovered that if an atom of uranium-235 is struck by a neutron (symbol n), the neutron initially sticks to the atom to make uranium-236. If left by itself it has a half-life of 7.1 x 10 8 years. Uranium-235, while being less stable than uranium-238, is still quite a stable atom. Likewise, if we left it for another 5730 years, half of the carbon we had left would have decayed and no longer be carbon, and so on: That is, if we had a jar of carbon-14 and left it on a shelf for 5730 years half of it would have decayed into another element (nitrogen-14) and we would be left with only half the original amount of carbon. For example, carbon-14 has a half-life of 5730 years. 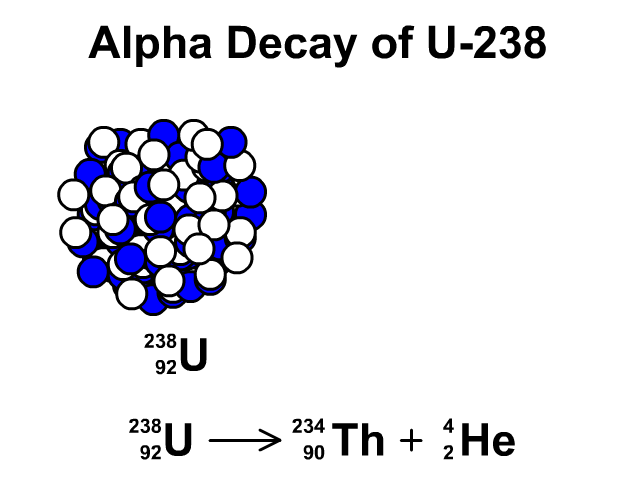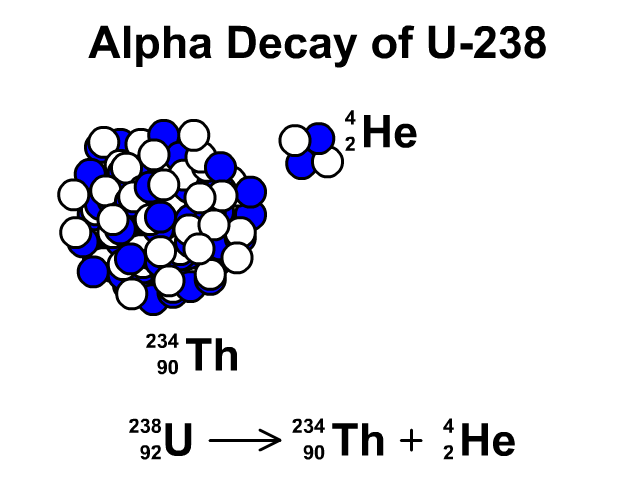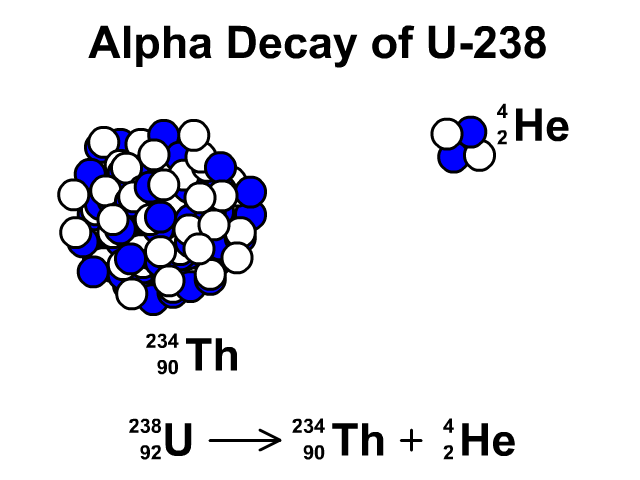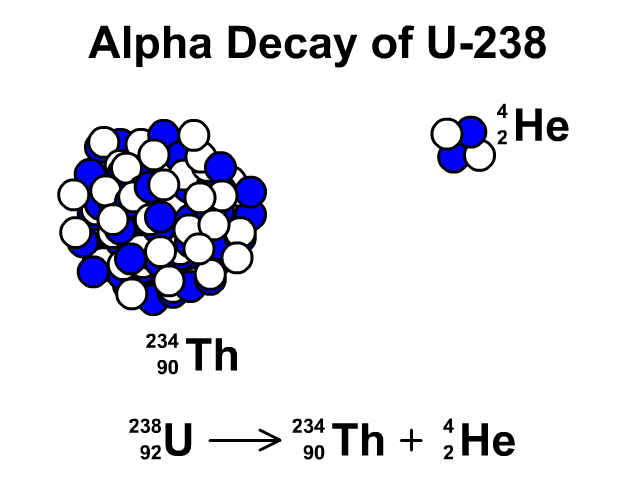
This is called the half-life of an element. We can't say when an individual atom will decay, but we can use statistical techniques to say how long it will be before a lot of atoms of a particular type decay to half of their original amount. Some of them take a very, very short time (a few billionths of a second) and some a very, very long time (possibly 10 32 years for hydrogen). All atoms decay, in other words, fall apart. Uranium-235 is also stable but undergoes radioactive decay a little more frequently. Uranium-238 is a stable isotope, that is, it only rarely undergoes any form of radioactive decay. In both cases they have 92 protons at their nucleus, and the bulk of each isotope is composed of neutrons. By far the most common are uranium-238 (99.3%) and uranium- 235 (0.7%). In the same way, there are different isotopes of uranium. However, in all cases carbon still has 6, and exactly 6, protons. Adding together the number of protons and neutrons gives us the isotope of the element, such as carbon-11 or carbon-14. For example, carbon has 6 protons, but can have different numbers of neutrons also within its nucleus. You notice that $57+42 \neq 92$, if that was the case, it would be equal, but I don't clearly see where the difference physically comes from and what to add or subtract (and why) from to the first or from the second term to get the other result.From previous pages in this series we know an element is defined by the number of protons in its nucleus. 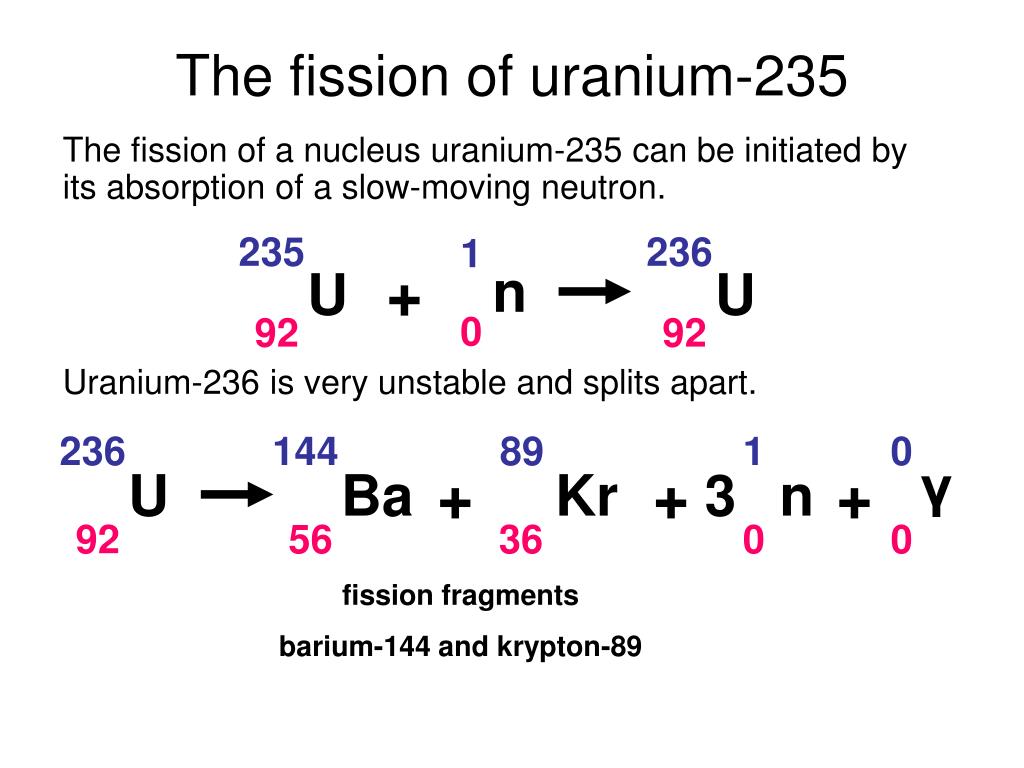 
Consider the neutron induced fission $\text 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
